 Magnesium has a valency of +2 and bicarbonate anion has a valency of -1. However, with ammonia: H N O X 3 ( a q) + N H X 3 N H X 4 N O X 3 How am I supposed to know this equation Its not metal + acid salt + water. 
So using the criss-cross method, you get Z n ( N O X 3) X 2 and water. In order to find the chemical formula using the criss-cross method, we have to know the valencies of the cation and anion. In the second part, for example, Z n is 2+ and H is -1. However, we will consider them as the magnesium cation and the bicarbonate cation. By seeing the compound name, we can see that the compound consists of magnesium and the bicarbonate. Now, we have been given with the compounds name as magnesium bicarbonate. For example, $$ represent two different but very similar chemical formula of water and hydrogen peroxide respectively, where hydrogen peroxide has an extra oxygen atom. The chemical formula helps us to identify what elements are present in a particular compound, and how many (means the atomicity of each element). 
Now, every compound has a chemical formula. Worksheets are Writing formulas key criss cross method, Ionicbondingandwritingformulasnamekey part use. We can start from this assumption: if a compound is neutral the algebraic sum of the oxidation numbers of his atoms must be zero ( point. In order to answer our question, we need to learn about the chemical formula and how it can be found out using the criss-cross method. Displaying all worksheets related to - Criss Cross Method. The resultant base formula is identical to what was derived using the first method: Al2S3. This interchanged valency numbers represent the atomicity of the element in the final chemical formula. The 'Criss-Cross' Method In this system, the superscript on the first symbol becomes the subscript on the second symbol, and the superscript on the second symbol is repositioned as the subscript on the first symbol, as shown below. Now, interchange the valencies with the other element. Metal + Non-Metal Ionic Compound If we have a transition metal the charge will be shown in parentheses after the element name. 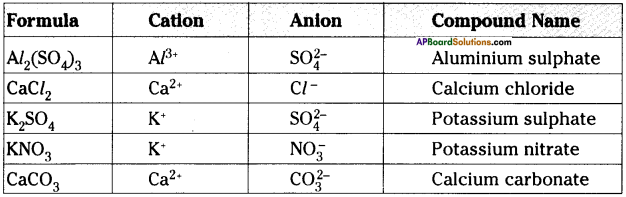
Hint Write down the name of the element and its valency below. Compounds with Transition Metals: Formula Writing When we have a metal and a non-metal we have an ionic compound. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
